Inhaled medication against pulmonary fibrosis
Hope for patients with pulmonary fibrosis: In an international study, researchers at LMU University Hospital Munich have shown for the first time that an inhaled drug can significantly slow down the progression of the severe lung disease and improve quality of life at the same time. The already known drug treprostinil, previously used for pulmonary hypertension, proved to be effective and well tolerated .
The results, which have been published in the New England Journal of Medicine, mark an important step forward in the treatment of idiopathic pulmonary fibrosis - a disease that has been difficult to treat until now. Further studies will now clarify whether the new therapy concept will soon be approved.

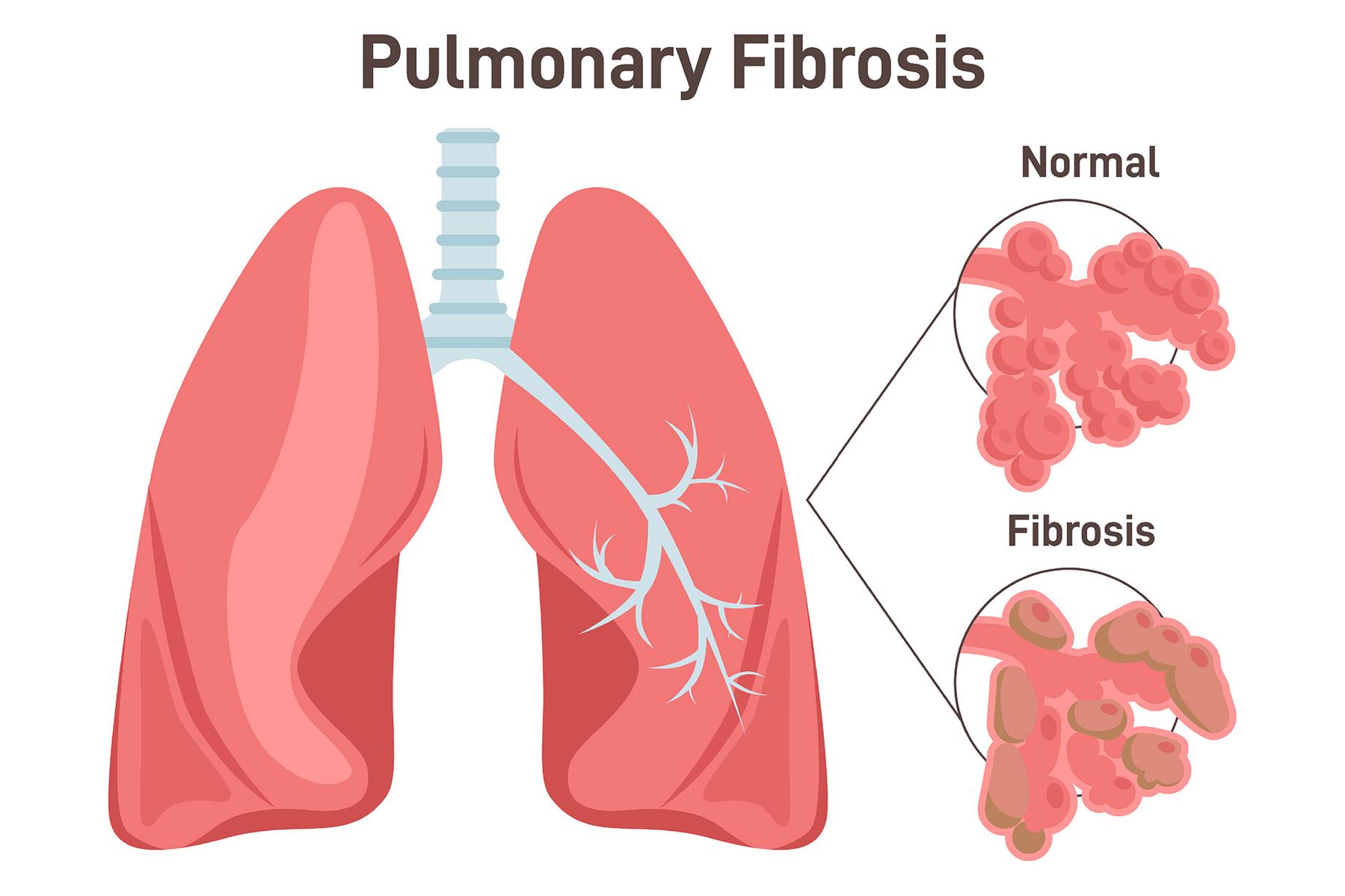
Idiopathic pulmonary fibrosis (IPF) is the prototypical fibrotic lung disease (synonym: interstitial lung disease, ILE), which is associated with a high mortality rate. In addition, there are over 200 causes of ILE, which in around a third of cases are also associated with scarring of the lung tissue, so that the inhaled oxygen can no longer be adequately passed on to the body.
As soon as scarring ("fibrosis") occurs, the consequences are comparable to idiopathic pulmonary fibrosis: increasing shortness of breath, dry irritating cough, decreasing performance and the need for oxygen supply through devices. "Many of the patients," says Jürgen Behr, "die within three to five years of the onset of the disease."
Previously available drugs can only slow down progression
Although IPF is rare in Germany, with 25 to 30 patients per 100,000 inhabitants, the incidence of the disease rises sharply, particularly in people over the age of 65. The drugs currently available can only slow down the progression of the disease and are associated with side effects.
A radical improvement in quality of life and survival can only be achieved through a lung transplant, but this is only an option for a small proportion of those affected. Not only in view of the shortage of donor organs, it is therefore essential that research into the disease continues unabated.
International study on the effect of inhaled treprostinil on pulmonary fibrosis
After the drug treprostinil has already been used successfully for years in patients with pulmonary hypertension, its effectiveness in patients with pulmonary fibrosis and additional pulmonary hypertension was investigated in a study. This study showed for the first time that inhaled treprostinil also has a positive effect on pulmonary fibrosis. On this basis, Munich physicians, together with colleagues from several international hospitals, conducted a large study to test whether inhaled treprostinil has a positive effect on idiopathic pulmonary fibrosis regardless of the presence of pulmonary hypertension.
The study was conducted in a double-blind, randomized and prospective format and included a total of 593 patients with an average age of 72 years. The forced vital capacity (FVC) was measured repeatedly over a period of one year. FVC is a measure of lung volume, which decreases rapidly in pulmonary fibrosis as a result of lung shrinkage.
Lung volume decreased less in the treprostinil group than in the placebo group
Result: The FVC of the participants in the placebo group decreased by a good 136 milliliters, while that of the participants in the treprostinil group only decreased by just under 50 milliliters. In this group, around 80 patients (27 percent) deteriorated clinically in terms of their symptoms during the study period, compared to 115 patients (39 percent) in the control group. "Survival without progression of the disease was also better in the treprostinil group," emphasizes Jürgen Behr.
Overall, the drug was well tolerated. Not unexpectedly, coughing during inhalation occurred in just under half of the patients in the treprostinil group and in just under a quarter of the patients in the placebo group. A third of the participants in the group with the real drug discontinued treatment due to side effects, compared to a quarter in the control group.
Quality of life in treprostinil patients developed better than in the control group
"Compared to the previous substances," summarizes Jürgen Behr, "this new drug promises better efficacy with a favorable side effect profile." He goes on to say that "with certain measures, we can get the cough under control in most patients".
He was particularly impressed by the fact that the quality of life of the participants developed clinically-relevant better under the therapy than in the control group. Whether the drug will be approved in Germany will be decided after the results of two further studies, which have not yet been completed.
FAQ: Pulmonary fibrosis and treprostinil
In pulmonary fibrosis, damage to the alveoli (air sacs in the lungs) leads to defective healing with the formation of scarred areas and shrinkage of the lungs.
Due to the loss of alveoli, oxygen can no longer be adequately transferred to the blood, which initially leads to a lack of oxygen during exertion and later also at rest, and performance decreases further and further.
At the same time, the lung tissue becomes increasingly stiff due to the scarring, so that the work of breathing increases and the subjective feeling of breathlessness develops.
First of all, possible causes must be found or ruled out. These include certain organic or inorganic dusts (e.g. quartz dust, asbestos, mold-contaminated dusts in agriculture, etc.). Medication and rheumatic diseases can also trigger pulmonary fibrosis. In these cases, avoiding or treating the triggering cause is crucial.
If fibrosis has already occurred, there are two approved drugs - nintedanib and pirfenidone, which delay the progression of the disease and, in particular, lung shrinkage. A third drug with a slightly more favorable side effect profile - nerandomilast - has already been approved in the USA and is expected to be available in Germany in 2026.
In addition, pulmonary fibrosis patients benefit from general and symptomatic treatment measures such as oxygen administration if oxygen deficiency is already present, pneumological rehabilitation and moderate exercise, good nutrition with avoidance of obesity as well as of weight loss.
If all conservative treatment options have been exhausted and the disease is still progressing, lung transplantation remains the definitive treatment option. However, this requires that the patient is neither too healthy nor too ill and is therefore only an option for a small proportion of those affected.
Treprostinil is a prostacyclin analog that represents a novelty in the field of pulmonary fibrosis therapy due to its inhaled administration. Due to its effect on the pulmonary vascular system and the connective tissue cells, it can have a doubly beneficial effect on the disease process in pulmonary fibrosis. The study data now available are therefore particularly encouraging, as the shrinkage process has stabilized and the quality of life and, in particular, breathlessness have improved compared to the control group.
Treprostinil was originally developed for the treatment of pulmonary hypertension and is approved for both subcutaneous and intravenous administration in this indication and is used very successfully. In the so-called INCREASE study, inhaled treprostinil was tested in patients with pulmonary fibrosis and additional pulmonary hypertension with positive results, as a result of which the US Food and Drug Administration (FDA) approved inhaled treprostinil for this indication. In Europe, however, approval is still pending.
In individual cases, Treprostinil can be imported via the international pharmacy (internationale Apotheke) and administered as "off label use". However, the organizational effort and costs are very high. It also requires individual approval and confirmation of cost coverage by the respective health insurance company.
Currently, two large studies with inhaled treprostinil are still being conducted for the indication idiopathic pulmonary fibrosis (IPF) in the USA and Canada and for the indication progressive pulmonary fibrosis (worldwide). If these studies confirm the positive results of the first study, approval can be expected.
Author
Prof. Dr. med. Jürgen Behr is a specialist in internal medicine, pulmonary and bronchial medicine, cardiology and allergology. He holds the Chair of Internal Medicine/Pneumology at Ludwig-Maximilians-Universität München (LMU), is Director of the Department of Medicine V (Pneumology) at LMU University Hospital Munich and a member of the Board of Directors of the Stiftung AtemWeg.
Publication
Inhaled Treprostinil for Idiopathic Pulmonary Fibrosis
Steven D. Nathan, M.D., Peter Smith, Pharm.D., Chunqin Deng, M.D., Ph.D., Maria De Salvo, M.D., Wim Wuyts, M.D., Juana Pavie-Gallegos, M.D., Jin Woo Song, M.D., Ph.D., +11 , for the TETON-2 Trial Investigators *Author Info & Affiliations
NEJM, Published March 11, 2026
Sources and further links
- Website of the Department of Medicine V (Pneumology)
- Video with Prof. Jürgen Behr: Pulmonary fibrosis - short and easy to understand
- S2k guideline on the diagnosis of idiopathic pulmonary fibrosis
- S2k guideline on pharmacotherapy of idiopathic pulmonary fibrosis (an update) and other progressive pulmonary fibrosis
- News report LMU University Hospital Munich: Pulmonary fibrosis: Ion channel regulates tissue remodeling
Contact
Prof. Dr. med. Behr, Jürgen
Director Department of Medicine V (Pneumology), LMU University Hospital Munich
